 Additional benefits associated with flash glucose monitoring versus SMBG include reductions in acute diabetes events, all-cause hospitalizations and hospitalized ketoacidosis episodes improved well-being and decreased disease burden and greater treatment satisfaction. Higher (versus lower) scanning frequency was associated with significantly greater reductions in HbA1c and significant improvements in other measures such as time spent in hypoglycemia, time spent in hyperglycemia, and time in range. Real-world studies in both type 1 diabetes or T2D populations also showed that flash glucose monitoring improved glycemic control. Subsequent RCTs and real-world chart review studies have since shown that flash glucose monitoring significantly reduces HbA1c from baseline. Compared to SMBG, the flash system significantly reduced the time spent in hypoglycemia and frequency of hypoglycemic events, although no significant change was observed in glycosylated hemoglobin (HbA1c) levels. In patients with type 2 diabetes (T2D) receiving insulin therapy, pivotal efficacy data were provided by the 6-month REPLACE randomized controlled trial (RCT) and 6-month extension study. Swiping the reader device over the sensor retrieves stored data and displays current interstitial glucose levels, a glucose trend arrow, and a graph of glucose readings over the preceding 8 h. 
The FreeStyle Libre flash glucose monitoring system (Abbott Diabetes Care, Witney, United Kingdom) consists of a factory-calibrated sensor worn on the back of the arm which measures glucose levels in the interstitial fluid every minute and stores the reading automatically every 15 min. FreeStyle, Libre and related brand marks are marks of Abbott.Continuous glucose monitoring systems are increasingly being adopted as an alternative to self-monitoring of blood glucose (SMBG) by persons with diabetes mellitus receiving insulin therapy. All trademarks referenced are trademarks of either the Abbott group of companies or their respective owners. For additional information, please read the user’s manual. The Sensor must be removed prior to Magnetic Resonance Imaging (MRI). This material is intended for Healthcare Professionals. Product is in Compliance with the applicable regulation on medical devices. Not all features are available in all markets. Product availability may vary by country. If you live in another country, please contact your Local Abbott office to obtain the correct product information for your country of residence.įreeStyle Libre Portfolio is available in both mmol/L and mg/dl versions depending on the country. This is a product specific website intended for Health Care Providers of the European Union only. Sensor is water resistant in up to 1 metre (3 feet) of water for a maximum of 30 minutes. 60-minute warm-up required when applying the sensor.Ĩ. 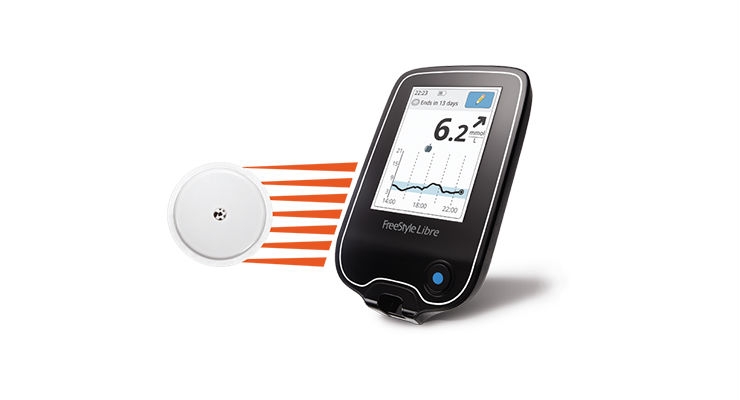
Dexcom G6 CGM User Guide and Medtronic Guardian Connect System User Guide.ħ. “Accuracy of a 14-Day Factory-Calibrated Continuous Glucose Monitoring System With Advanced Algorithm in Pediatric and Adult Population With Diabetes.” Journal of Diabetes Science and Technology, (September 2020).Ħ. Christiansen, Gregory Forlenza, Mark Kipnes, David R. Alva, Shridhara, Timothy Bailey, Ronald Brazg, Erwin S. Data on file, Abbott Diabetes Care, Inc.ĥ. 
Use of FreeStyle LibreLink requires registration with LibreView.Ĥ. Please check the website for more information about device compatibility before using the app. The FreeStyle LibreLink app is only compatible with certain mobile devices and operating systems. Once the patient scans their FreeStyle Libre 2 sensor with that device, they can receive alarms only on that device. They must start their FreeStyle Libre 2 sensor with that selected device. Patients choose which device they want to receive alarms: FreeStyle Libre 2 reader or FreeStyle LibreLink app. Finger pricks are required if your glucose readings and alarms do not match symptoms or expectations.ģ. 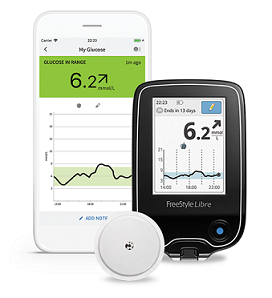
Data based on the number of users worldwide for the FreeStyle Libre system compared to the number of users for other leading personal use sensor based glucose monitoring systems.Ģ. Images are for illustrative purposes only. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
